
How Scientists Are Trying to build Mini-Organs for the Masses
Imagine testing a life-saving drug not on an animal, but on a miniature, lab-grown replica of a human organ, i.e. ‘organoid.’ This technology, which uses human stem cells to create tiny, self-organizing 3D tissues, is arguably the biggest leap in biomedical science in decades.
Organoids, such as mini-livers, mini-kidneys, or even tiny tumors, offer a level of biological accuracy that was previously impossible. They hold the key to faster drug discovery and, potentially, truly personalized medicine.
So, if the technology is so revolutionary, why aren’t pharmaceutical companies using them to screen every new drug candidate?
This brings us to the core challenge addressed by a new patent from Molecular Devices UK, US12351820B2: the problem of turning a scientific miracle into an industrial reality.
A Scientific Leap Trapped in a Small Lab
For years, growing organoids has been more of an art than a science. The process relies heavily on the technical skill of a specialized lab technician, resulting in a culture system that is fundamentally unreliable and unscalable.
- Low Production, High Effort: Organoids are typically grown in small batches in a restrictive gel matrix. This process is time-consuming and yields small numbers, making them unsuitable for High-Throughput Screening (HTS), where millions of compounds must be tested quickly.
- The “Snowflake” Problem: The most significant issue is a lack of standardization. Since the process is manual, the resulting organoids vary wildly from batch to batch, and even within the same batch. They are like snowflakes, each one is unique. This high variability is a nightmare for drug testing, where results must be consistent and repeatable to be valid. You can’t trust the results if the ‘testing grounds’ keep changing.
The pharmaceutical industry needs a process that can consistently produce thousands of identical, high-quality organoids for drug testing and disease modeling. This patent proposes the blueprint for that industrial-scale solution.
A Three-Step Recipe for Consistency
The patent’s core invention is a three-step method to manufacture homogenous (uniform) organoids at a massive scale in automated bioreactors.
- Dissociation and Breakdown: First, existing organoids are chemically or mechanically broken down into a suspension of individual cells and small cell clusters. This is the raw material for the next batch.
- The Critical Standardization Step (Sieving): This is the game-changer. The cell suspension is passed through a cell strainer, a specialized, fine-tuned sieve. This filtration step removes all the irregular, too-large, or too-small cell clusters by selecting a very narrow and specific size range (e.g., between $10 \mu\text{m}$ and $1 \text{mm}$). This precise size selection ensures that every new organoid starting point is identical, which, in turn, guarantees the final organoids will be uniform in size and quality.
- Industrial Scale-up (Bioreactor Culture): Finally, the now-standardized cell clusters are immediately introduced into a bioreactor—a large, controlled vessel, often a ‘fed-plate’ or ‘flat-bed’ type. The bioreactor automates the supply of nutrients, oxygen, and growth factors for continuous, high-density cultivation that yields mass production of consistent, high-quality organoids.
Why It Matters
The idea of growing organoids in large-scale bioreactors (like stirred-tank or rotating systems) is an active area of research. It’s the obvious path to scale up volume, and other patents and technologies are in this space.
However, the key novelty of this Molecular Devices UK patent is the mandatory insertion of the size-selection (sieving) step immediately before the bioreactor culture.
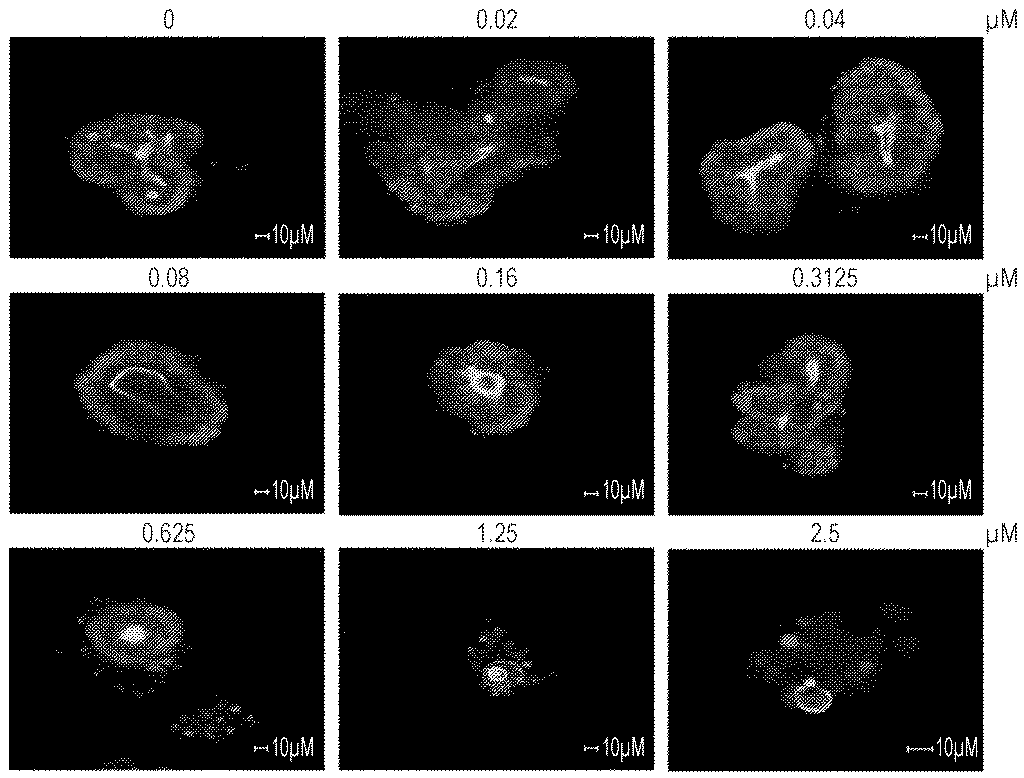
While existing solutions focus primarily on volume (how many can we grow?), this patent uniquely tackles the industrial-scale challenge of consistency (how many identical ones can we grow?). Any number of bioreactors can grow cells quickly, but if the final product is variable, it’s useless for pharmaceutical screening.
By integrating the physical sieving of cell clusters as a required precursor to the automated, high-volume growth phase, the patent provides a method for predictable, high-quality, and standardized mass production, which is a critical distinction in the race for industrial dominance.
Industry Impact and Business Implications
If this three-step process becomes the industry standard for reliable organoid manufacturing, its effect on the biomedical world will be profound:
- Accelerating Drug Discovery: The standardized, high-quality product removes the biggest barrier to using organoids in High-Throughput Screening (HTS). Pharmaceutical companies can use these mini-organs to test millions of drug compounds with unprecedented accuracy, rapidly identifying successful candidates and predicting human toxicity before expensive and often misleading animal trials. This translates directly to faster development timelines and billions saved in failed clinical trials.
- The Rise of Personalized Medicine: This method makes the routine, large-batch production of Patient-Derived Organoids (PDOs) feasible. A doctor could take a biopsy from a patient’s tumor, grow hundreds of identical mini-tumors in the bioreactor, and then rapidly test dozens of different drugs to determine which one works best for that specific patient.
- Automation and Cost Reduction: By moving the complex, sensitive process from a highly-skilled manual procedure on a lab bench to an automated, standardized bioreactor system, the method dramatically lowers labor costs and increases manufacturing consistency, making organoid technology a financially viable commodity for research and clinical applications. This patent provides the technical foundation for the organoid to move from a scientific curiosity to a core industrial tool.
Conclusion
By combining the crucial size-selection (sieving) step with automated bioreactor culture, Molecular Devices UK has provided a blueprint to standardize the production of these complex 3D tissues.
This shift from manual craft to industrial process removes the main bottleneck in adopting organoids for drug development. The successful scaling of this technology promises to usher in an era of faster, more accurate drug screening and finally make highly precise, personalized medicine a routine clinical reality.



