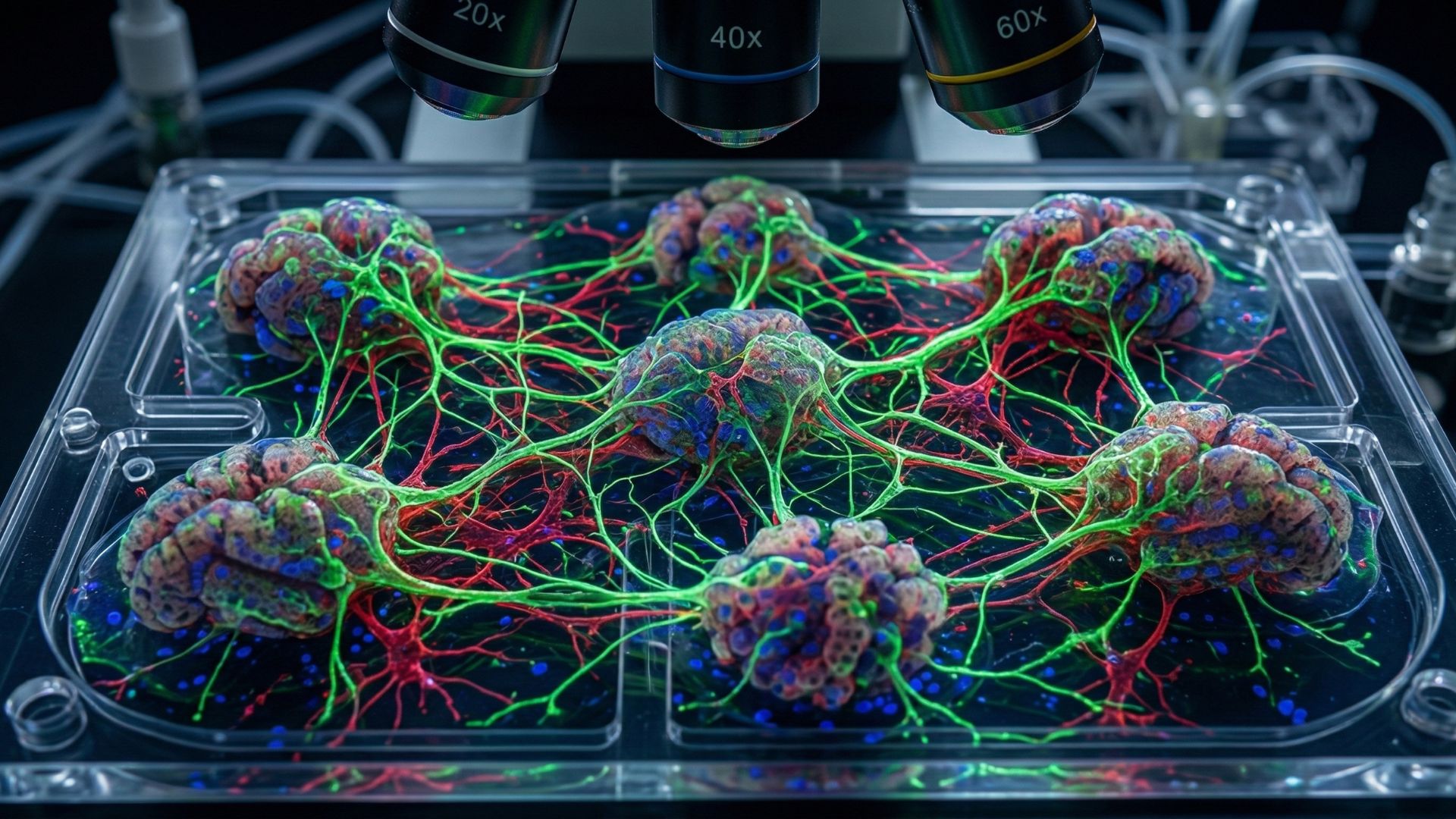
What if the future of medicine and computing wasn’t built in factories, but grown in dishes? Imagine tiny clusters of cells arranging themselves into miniature organs, or networks of living neurons firing like circuits. These aren’t scenes from science fiction anymore; they’re real experiments happening in this world.
Scientists call them Organoids and Wetware, and together they blur the line between biology and technology. Organoids are tiny, lab-grown versions of human organs made from stem cells. They are a category of stem cell-derived three-dimensional (3D) cell aggregates that partially emulate the developmental trajectory of natural organs as well as the tissue-specific structure and function.
Over the decades, computing has been defined by silicon chips, transistors, and binary code. But now a new frontier is emerging- one that doesn’t rely on metal or plastic, but on living cells. It’s the Wetware i.e “biological hardware + software” that enables thought, memory, and consciousness.
Wetware is the living circuits made of neurons pulsing with electrical activity. These are the two breakthroughs that are redefining how we study disease, design medicine, and even imagine computing
In computing terms, neurons act like hardware, and signals act like software, and wetware is the whole living system. Wetware uses living cells- like neurons- as circuits, creating “biological computers” that can think and adapt in ways machines cannot. The merging of organoids (miniature lab-grown organs) with wetware marks a groundbreaking step in technology, turning living cells into active, energy‑efficient tools for both medical breakthroughs and new forms of computational intelligence.
In this article, we will explore what exactly is Organoid and how it is disrupting human lives.
What are Organoids?
Organoids are miniature, simplified versions of human organs grown in 3D culture from stem cells, designed to mimic the structure and function of real organs. (3D) structures are derived from stem cells, which can be either pluripotent (capable of becoming any cell type) or adult (tissue-specific) stem cells.
Organoids, such as mini-livers, mini-kidneys, or even tiny tumors, offer a level of biological accuracy that was previously impossible. This technology, which uses human stem cells to create tiny, self-organizing 3D tissues, is the most groundbreaking leap in biomedical science in decades.
These miniature organs are smaller than their biological counterparts, measuring a few millimeters across. Organoids are not fully developed organs as they lack the comprehensive network of blood vessels, immune systems, and the complexity of whole-body integration, but they bridge the gap between oversimplification involving growing animal or plant cells in a controlled, artificial environment after isolating them from tissues.
Core Technologies Behind Organoid
1. Stem Cell Engineering
Stem cell engineering is the foundation on which Organoid technology is built.It is the science of deliberately designing, modifying, and controlling stem cells to achieve precise biological outcomes.These cells have a remarkable ability to turn into almost any tissue in the body and have the capacity to differentiate into nearly any tissue within the human body.
Advancement in stem cell research is playing a major role in reprogramming ordinary adult cells, opening the door to breakthroughs in regenerative medicine and personalized healthcare.Together, these innovations lay the foundation for a new era in healthcare- one in which treatments are not simply prescribed, but cultivated from living cells.
2. Extracellular Matrix (ECM) & Bioengineering
The Extracellular Matrix (ECM) is the material that surrounds our cells. You can think of it like a soft scaffold or support system that holds cells in place and helps them talk to each other. It is made up of proteins and sugars that guide how cells grow, move, and repair themselves.
Advancements in bioengineering are revolutionizing healthcare by enabling scientists to grow new tissues and organs, guide stem cells to become specialized cells, deliver medicines with precision, and recreate disease conditions in the lab for better understanding and treatment.
By working with the extracellular matrix (ECM), researchers can rebuild damaged organs, create treatments designed for each person, and better understand complex illnesses. This fusion of ECM biology and bioengineering is opening the door to a new era of regenerative medicine—one where the body’s own blueprint is used to engineer cures.
3. Microfluidics (Organ-on-a-Chip)
Microfluidics is a technology that combines micro-manufacturing and tissue engineering to replicate the physiological environment and functions of human organs on a microchip. These devices are typically the size of a USB stick and contain hollow channels lined with living human cells, allowing for real-time monitoring of organ-level responses to drugs and diseases.
Organ-on-a-chip is a cutting-edge technology that enables performing more accurate research on human physiology and initiates advancements in artificial disease models. Among them, we can find models like gut-on-a-chip, heart on a chip, muscle on a chip, multiple organs on a chip, etc.
4. Advanced Genetic & Computational Integration
Gene Editing
Gene editing is the precise modification of DNA to add, remove, or alter genetic material, with CRISPR-Cas9 being the most widely used tool today. When gene editing combines with organoids, it creates one of the most transformative platforms in modern science—tiny, lab-grown organs that can be precisely engineered to model disease, test therapies, and even explore futuristic technologies.
Organoid Intelligence (OI)
The vision behind Organoid Intelligence (OI) is to cultivate miniature brain-like structures in the laboratory and train them to process information like a real human brain, utilizing neurons. Organoid Intelligence is still in its early days, but its possibilities are enormous, and it could change both medicine and technology. Imagine computers that learn like brains, or lab-grown tissues that help cure diseases – that’s the promise of OI.
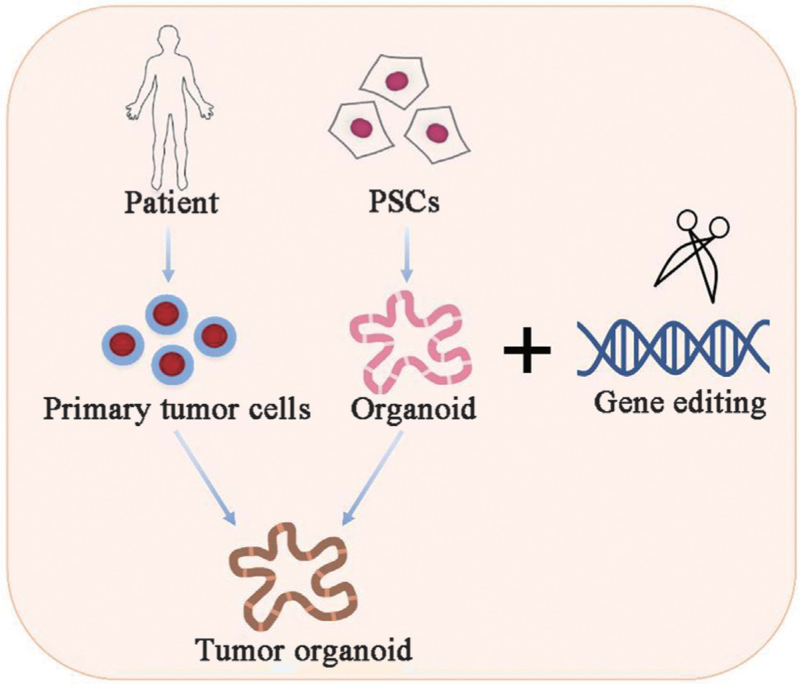
Advantages of Organoids
1. Cancer Research
Cancer is a very complex disease, and for studying, we need models that can show how real tumours behave. Traditional methods like flat cell structures were used, but often they miss the important details.
But as technology has progressed, Organoids preserve the genetic changes of a patient who is suffering through cancer, giving a more realistic model to study how tumors develop, spread, and respond to treatment.
Drug testing is carried out on realistic cancer models called organoids, and this research supports personalized therapy selection for individual patients. By using organoids, doctors can test different drugs directly on a patient’s tumor model to see which treatment is most effective.
2. Genetic Disorders
A genetic disorder is a disease caused by mutations in a person’s DNA. These mutations can be inherited from parents or occur naturally. As organoids are made of human stem cells, they carry the same genetic defects as the patient, allowing the scientist to study how these mutations affect organ growth, function, and disease development. A safe platform is developed by Organoid to test new therapies, including advanced gene-editing tools like CRISPR, to see if faulty genes can be corrected.
3. Neurological Diseases
Neurological diseases are disorders that affect the brain, spinal cord, and nerves. Brain organoids are transforming the landscape of neurological disease research. By providing human-like three-dimensional models, they replicate aspects of brain development and pathology far more effectively than traditional two-dimensional cultures or animal models. They open new avenues for understanding, diagnosing, and treating some of the most challenging conditions in medicine.
To study these disorders, organoids help in studying neurological disorders such as Alzheimer’s disease, Parkinson’s disease, and Autism spectrum disorders in a realistic way to understand how they develop and explore new treatments.
5. Drug Resistance & Precision Therapy
Drug resistance is one of the biggest challenges in modern medicine, and precision therapy offers a way to overcome it by tailoring treatments to the unique genetic, molecular, and cellular features of each patient’s disease.
Limitations of Organoids
1. Lack of Maturity & Function
Organoids are still in their developing stage, and their lack of maturity and full functionality is one of the biggest hurdles. Organoids often remain in fetal or immature states, lacking the full cellular complexity and function of adult organs, like proper blood vessels or complex nerve connections.
2. No Vascularization
Vascularization is the missing piece that limits organoid growth and functionality. Without blood vessels, organoids remain small, short-lived, and unable to fully mimic real organs. In the human body, blood vessels are the lifelines of every organ. They deliver oxygen, nutrients, hormones, and remove waste.
Without them, no tissue can grow beyond a certain size. But in the case of Organoid blood vessels, nutrient and oxygen supply is limited, which restricts organoid size and long-term survival, as cells in the core can’t get enough resources.
3. Variability & Heterogeneity
Variability in organoids means differences in size, shape, cellular composition, or function among organoids derived from the same protocol or cell source. Organoids grown from the same stem cell line can differ in size, shape, and maturity. Some may develop more cortical layers, while others remain rudimentary.
Heterogeneity in organoids is diversity in cell types, states, or structures within a single organoid or across different organoids. Within a single organoid, you may find different neuronal subtypes (excitatory, inhibitory), glial cells, and progenitors. Across organoids, some resemble fetal-like tissue, while others show more adult-like features.
4. Slow Development
Organoids develop slowly, often taking weeks to months to grow. This extended timeline makes them impractical for urgent clinical decisions, such as guiding first-line cancer treatments where speed is critical.
5. Unclear Microenvironment Influence
The role of the microenvironment in organoid development remains uncertain. Extracellular matrix materials such as Matrigel can influence cell behavior, raising questions about whether experimental outcomes truly reflect natural tissue conditions.”
Ethical Barriers in Organoids
1. Informed Consent and Donor Rights
Organoids are typically derived from human stem cells or tissue donations. Donors often face a significant gap in understanding how their cells will be ultimately used, especially if research evolves into commercial applications.
Ensuring transparency and consent of patients are the important factors in this process that create ethical and legal barriers in organoid research. When donors provide stem cells or tissue samples, they often agree to their use for scientific study without fully realizing that those cells could later be employed in drug testing, transplantation, or even commercial ventures.
The challenge lies in guaranteeing that consent is not a one-time event but a continuous process that adapts as research evolves. This raises serious questions of autonomy, fairness, and justice, highlighting the need for legal frameworks that protect donor rights while promoting scientific innovation.
2. Consciousness in Cerebral Organoids
Cerebral organoids, often referred to as “mini-brains,” present one of the most complex ethical challenges in organoid research. These structures can mimic aspects of neural development and, in some cases, exhibit electrical activity resembling brain waves.
The scientific community remains divided: some argue that current organoids are far too rudimentary to support consciousness, while others caution that advancing technologies could eventually cross this threshold.
This debate raises profound ethical alarms, demanding careful oversight, clear guidelines, and ongoing dialogue to ensure that scientific progress does not inadvertently create entities with moral status that society is unprepared to recognize or protect.
3. Clinical Translation and Patient Safety
Clinical translation and patient safety form another critical barrier in organoid research. While organoids hold immense promise for regenerative medicine- such as repairing damaged tissues or replacing failing organs-their direct application in patients raises significant risks.
These include the potential for tumor formation, immune rejection, and unpredictable long-term effects once transplanted into the human body. The ethical dilemma lies in balancing the urgency of medical innovation with the responsibility to protect patients from harm.
Researchers and regulators must ensure that rigorous testing, transparent clinical trials, and strict oversight accompany any move toward clinical use. Without such safeguards, the rush to translate organoid science into therapies could compromise patient safety and undermine public trust in biomedical innovation..
4. Commercialization and Ownership
Commercialization and ownership present a significant barrier in organoid research, as the line between scientific discovery and profit-driven enterprise is often blurred. Organoids, once developed from donated human cells, can be patented and commercialized, raising difficult questions about who truly owns the biological material and who should benefit from its use.
Donors may unknowingly contribute to therapies or products that generate substantial profits without receiving recognition or compensation, creating concerns of exploitation and inequity. At the same time, companies and research institutions argue that intellectual property rights are essential to incentivize innovation and secure funding for further development.
This tension highlights the need for legal frameworks that balance the protection of donor rights with the realities of commercial investment, ensuring that organoid science advances responsibly without reducing human biological contributions to mere commodities.
5. Equity and Global Access
Equity and global access represent one of the most pressing barriers in organoid research, as the benefits of these scientific breakthroughs risk being concentrated among wealthy individuals and nations. Organoid-based therapies, from personalized drug testing to regenerative medicine, are likely to be costly and resource-intensive, making them inaccessible to marginalized populations and low-income countries.
This raises serious ethical concerns about widening healthcare disparities and creating a divide between those who can afford cutting-edge treatments and those who cannot. Ensuring equitable access requires deliberate policies that promote affordability, international collaboration, and fair distribution of medical advances.
Without such measures, organoid science could inadvertently reinforce global health inequalities rather than alleviate them, undermining its potential to serve as a universal tool for human well-being.
6. Reproductive and Developmental Organoids
Reproductive and developmental organoids introduce some of the most sensitive ethical and legal challenges in this field. These organoids, derived from placental, embryonic, or reproductive tissues, are designed to model aspects of early human development, maternal-fetal interactions, or reproductive biology.
While they offer valuable insights into infertility, pregnancy complications, and developmental disorders, they also blur the line between scientific research and human reproduction. The barrier lies in the moral implications of creating structures that resemble embryonic or reproductive systems, raising questions about whether such organoids could be considered forms of life or whether they might one day be misused in reproductive contexts.
Legal Barriers in Organoid Research
1. Intellectual Property and Ownership
Organoids derived from donated cells can be patented, which raises complex disputes over intellectual property and ownership. The central issue lies in determining who truly owns the biological material once it has been transformed into organoids—whether it is the researchers, institutions, or the original donors.
This debate extends to whether donors should share in the profits generated from such patents or receive recognition for their contribution. As a result, questions of fairness, consent, and benefit-sharing emerge, highlighting the ethical and legal challenges surrounding the commercialization of human-derived biological materials.
2. Regulatory Oversight
Regulatory oversight in the field of organoid-based therapies remains a significant challenge, as laws often lag behind rapid scientific innovation. The absence of clear and comprehensive frameworks for ethical review, clinical trials, and commercialization creates uncertainty for researchers, clinicians, and companies working in this area.
Without well-defined guidelines, it becomes difficult to ensure patient safety, maintain ethical standards, and establish pathways for bringing organoid technologies from the laboratory to clinical practice.
This regulatory gap highlights the urgent need for updated policies that can keep pace with scientific progress while safeguarding public trust and responsible innovation.
3. Data Privacy and Genetic Information
Organoid research often involves handling highly sensitive genetic information, which raises significant concerns about data privacy and security. Because genetic data can reveal deeply personal details about individuals, researchers must adhere to strict data protection regulations to safeguard this information.
In Europe, the General Data Protection Regulation (GDPR) sets rigorous standards for how such data can be collected, stored, and shared, while in the United States, the Health Insurance Portability and Accountability Act (HIPAA) governs the protection of medical and genetic records.
Compliance with these frameworks is essential to ensure that genetic information used in organoid studies remains confidential, secure, and ethically managed.
4. International Disparities
Different countries have varying regulations on stem cell and organoid research, which creates the risk of “ethics tourism,” where researchers exploit weaker laws abroad to bypass restrictions. This disparity raises concerns about fairness and oversight in scientific progress.
At the same time, liability and patient safety become critical issues, as organoid-based therapies may cause harm such as tumor formation or immune rejection. In such cases, questions arise about who bears legal responsibility.
To ensure patient protection and guide clinical translation effectively, clear liability frameworks are essential, providing accountability while fostering trust in these emerging medical technologies..
Conclusion
Organoid research is full of promise. It can change medicine in amazing ways, but it also raises big questions about ethics and fairness. To make sure this science helps people without crossing moral lines, we need strong rules, honest communication, and teamwork across countries. If we balance progress with responsibility, organoid research can truly benefit humanity.